The Fact That No Two Electrons in an Atom Can Have the Same Set of Quantum Numbers Is Known as the
Pauli Exclusion Principle
- Folio ID
- 660
The Pauli Exclusion Principle states that, in an cantlet or molecule, no two electrons can have the aforementioned iv electronic quantum numbers. Every bit an orbital can contain a maximum of only two electrons, the two electrons must take opposing spins. This ways if ane electron is assigned as a spin up (+ane/ii) electron, the other electron must be spin-downwardly (-1/ii) electron.
Electrons in the aforementioned orbital take the same kickoff three quantum numbers, e.g., \(n=1\), \(l=0\), \(m_l=0\) for the anes subshell. Simply two electrons tin take these numbers, and so that their spin moments must be either \(m_s = -1/ii\) or \(m_s = +1/2\). If the 1s orbital contains only one electron, nosotros have one \(m_s\) value and the electron configuration is written as anes 1 (respective to hydrogen). If it is fully occupied, nosotros have two \(m_s\) values, and the electron configuration is 1southward 2 (corresponding to helium). Visually these two cases can be represented every bit
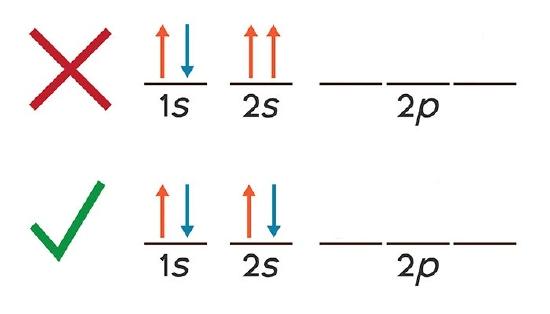
As you can see, the is and 2s subshells for beryllium atoms can hold only 2 electrons and when filled, the electrons must accept opposite spins. Otherwise they will have the same four quantum numbers, in violation of the Pauli Exclusion Principle.
Contributors and Attributions
- Sarah Faizi (University of California Davis)
-
Dr. Craig Fisher (Japan Fine Ceramics Center)
Source: https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textbook_Maps/Supplemental_Modules_(Physical_and_Theoretical_Chemistry)/Electronic_Structure_of_Atoms_and_Molecules/Electronic_Configurations/Pauli_Exclusion_Principle
0 Response to "The Fact That No Two Electrons in an Atom Can Have the Same Set of Quantum Numbers Is Known as the"
Post a Comment